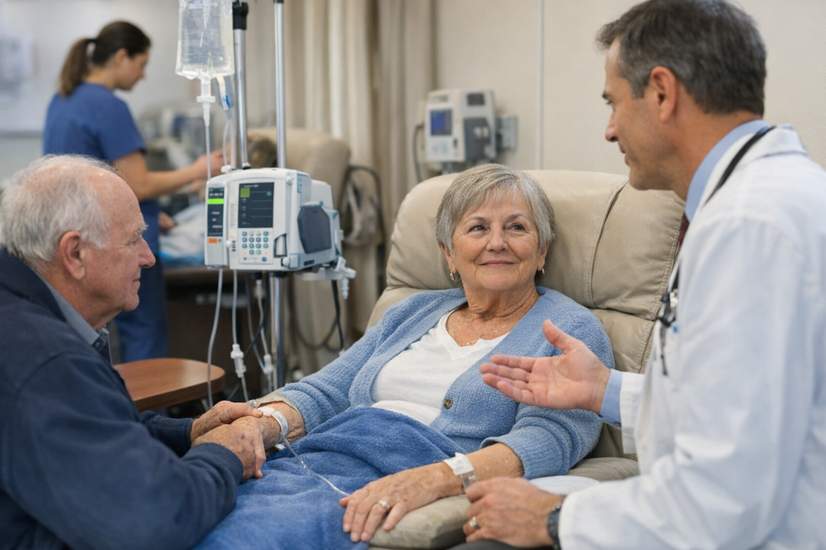Why everyone’s talking about immunotherapy (and why it doesn’t sound like a horror movie)
For decades, cancer treatment felt like a binary choice: cut it out, poison it, or scorch it. Lately, though, a very different playbook has been showing up in clinics—one that trains your own immune system to spot and attack tumors. The results can be dramatic, sometimes borderline sci-fi, and for many patients they’ve translated into real, lasting remissions with fewer of the classic chemotherapy side effects.
A patient story that reads like a plot twist
Take Maureen, a 71-year-old who’d faced colon cancer years ago with surgery and a tough recovery. When she developed a different cancer later in life, instead of another operation she tried a new immunotherapy infusion every few weeks. Within a few months, the tumor was gone—no scalpel, no radiation, just a handful of clinic visits and a manageable lingering fatigue from hormone changes. It’s the kind of comeback that makes doctors and patients do a double take.
What’s actually going on inside your body?
Your immune system is already on patrol, quietly policing cells that look “off.” But cancer can be sneaky—masking itself so immune cells don’t recognize it as a threat. Modern immunotherapy’s job is basically to rip off the disguise, hand the immune system a wanted poster, and say, “Go get ’em.” When it works, the body does what it was built to do: hunt and destroy abnormal cells.
The popular tools: CAR T and checkpoint inhibitors (in plain English)
Two headline-grabbing strategies are CAR T-cell therapy and checkpoint inhibitors. CAR T is like recruiting soldiers from your blood, giving them specialized training in the lab, and sending them back in to find cancer cells—super effective for some blood cancers. Checkpoint inhibitors, on the other hand, cut the brakes on immune cells so they can keep fighting; they’ve been revolutionary across multiple tumor types.
Why these treatments aren’t magic bullets (yet)
Both approaches have downsides. CAR T has been tougher to adapt for solid tumors and can be expensive and complex to produce. Checkpoint blockers can unbalance the immune system and cause inflammation in healthy organs—skin rashes, diarrhea or, more rarely, issues in the heart, liver or kidneys. And crucially, not everyone benefits: overall response rates tend to sit somewhere in the low-to-mid tens of percent, so many patients face side effects without much gain.
Thinking like a hacker: multipronged strategies to beat resistance
Researchers aren’t giving up. They’re trying everything from diet tweaks (hello, fiber fans) to re-purposing cheap drugs like statins, and even playing with the timing of doses. Pairing immunotherapy with radiation or focused ultrasound can help by making the tumor more visible to immune cells. In short: scientists are stacking and tuning therapies like a DJ mixing tracks, trying to find combos that get the tumor dancing to the immune system’s tune.
Personalized approaches: treating the person, not just the tumor label
Cancer is messy and diverse—two people with the “same” cancer can have very different diseases under the microscope. That’s why precision matchmaking between a patient’s tumor profile and the right immunotherapy is so exciting. In one notable trial, patients with a particular genetic fingerprint saw dramatic responses—many who completed treatment had tumors vanish. That opens the door to treatments that spare surgery and long recoveries, at least for a slice of patients.
Vaccine-style treatments: training your immune system on purpose
Think vaccines, but for an existing tumor: researchers can identify proteins on a person’s cancer cells and design a vaccine to prime the immune system to recognize and attack them. Early small trials—like a personalized vaccine given after kidney-cancer surgery—show people can mount targeted immune responses and stay disease-free for years. It’s early, but the idea of quickly developing a tailor-made vaccine against your specific tumor is thrilling.
Reality check: big promise, real limitations
There’s cautious optimism everywhere—but also caution. Only a minority of tumors carry the exact markers that current, surgery-avoiding immunotherapies target, and some tumors may be fundamentally resistant to any immune strategy. Many promising ideas stall in early trials, and more research is needed to match the right approach to the right patient. In short: progress is real, but it’s not a universal cure—yet.
Why this still feels like progress
For those who do respond, immunotherapy can be life-changing—turning invasive surgery and months of chemo into a relatively simple course of infusions or a targeted vaccine. Patients and researchers are hopeful that as we learn more about tumor fingerprints, the microbiome, timing, and smart combinations, the proportion of people who benefit will keep climbing. As one patient put it, the old ways of treating cancer may soon feel as outdated as bloodletting—dramatic, but in a good way.